AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Atomic mass of copper12/28/2023 
Mg-25 has a mass of 24.99 amu and accounts for 10.00% of natural magnesium. Mg-24 has a mass of 23.99 amu and accounts for 78.99% of all natural magnesium. Magnesium (Mg, element 12) has three natural isotopes: Mg-24, Mg-25, and Mg-26. This example will show how to find the average atomic mass of an element when given the natural abundance of each of the element’s isotopes. How to Calculate Atomic Mass From Natural Abundance Example These abundances are used to calculate the atomic mass value found on the periodic table. Copper-63 accounts for just under 70% of all copper found in nature. The average mass of copper takes account of the natural abundance of each isotope of an element. Copper-65 has two additional neutrons than copper-63 and therefore has more mass. The average is important since there are two different natural isotopes of copper: copper-63 and copper-65. This means the average mass of a mole of copper atoms is 63.546 grams.The average is important since there are two different natural isotopes of copper: Copper-63 and Copper-65. This value is the average atomic mass of the element because elements may have more than one naturally occurring isotope.Įxample: Find the element copper (Cu or element number 29) on the periodic table. The atomic mass for each element is given in atomic mass units or grams per mole of atoms. The easiest way to find the atomic mass is to look it up on a periodic table. For a single atom, this is the mass number, but for an element, it is the average atomic mass. This means the atomic mass is the sum of the masses of the protons and neutrons in an atom. 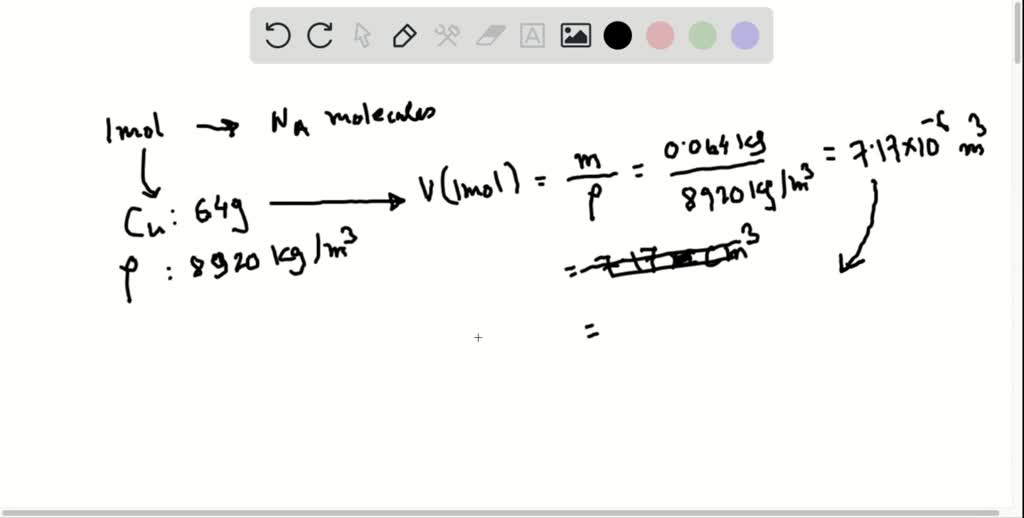
Electrons have nearly 1/2000th of the mass of protons and neutrons, so electrons are typically ignored in calculations involving atomic mass. Calculate the atomic mass of the elements boron and magnesium.Ītomic mass is the combined mass of all the protons, neutrons, and electrons that make up an atom.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
